Rare Earth Element Separation Using Mesoporous Materials
A simple and more efficient method for separating rare earth elements using mesoporous materials that eliminates hazardous solvents and complex, multi-step processes
Rare earth elements (REEs) possess unique optical and magnetic properties that are critical for next-generation electronics and emerging technologies. In contrast with trace metals such as copper and lead, REEs occur as dispersed mixtures in the earth’s crust that require separation of individual elements. Current separation techniques are complex and require multiple solvent extraction steps that produce large amounts of waste and must be fine-tuned for each new REE deposit. Due to rising global demand for REEs, there is a pressing need for simpler, more effective, and environmentally-friendly strategies for separating individual REEs, including those from low-concentration sources, such as geothermal fluids or waste streams.
Sandia researchers have developed a promising new method for separating individual REEs from aqueous mixtures using mesoporous materials such as silica and alumina. The innovation utilizes an important systematic variation in the adsorption affinity of REEs, where the affinity increases as the elemental mass increases, and pore diameter can be used to tune the adsorption affinity of mesoporous materials. Researchers demonstrated that one or more REEs can be separated from an aqueous mixture using a flow-through or column method. This development has potential to dramatically streamline industrial refining and separation of REEs by offering a simpler and more efficient method that eliminates the need for hazardous solvents and complex, multi-step processes.
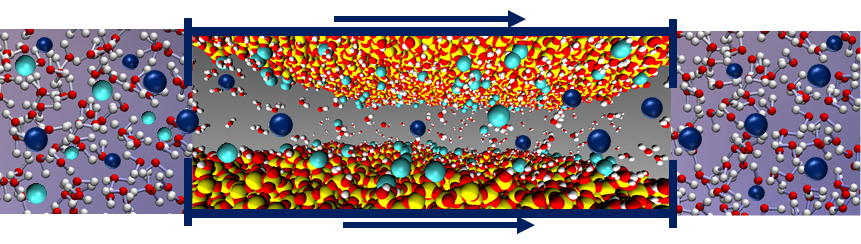
- Simple, single-step separation process for individual REEs
- Requires no hazardous solvents
- Useful for the separation of REEs from low-concentration sources, such as hydrothermal fluids or waste streams
- Stable in acidic solutions and useful for REE-enriched leachates
- May be extended to separation of actinides
- Mining and REE extraction
- Refining
- Separation science
SD# 14618
Published7/14/2020
Last Updated7/14/2020