Synthesis of Nanocrystalline Iron Nitrides Using Two-Step Reactive Milling Process
Nanocrystalline iron nitride is an important soft magnetic material; however, conventional methods of production don’t exist. Synthesis of dense nanocrystalline iron nitrides is not possible by simply annealing elemental iron in NH3 at temperatures in excess of 600° C since g’-Fe4N and other iron nitrides are unstable above 600°C and will decompose. Sandia researchers have discovered that by using a two-step reactive milling process and high pressure spark plasma sintering (SPS) they can quickly and efficiently fabricate bulk g’-Fe4N parts.
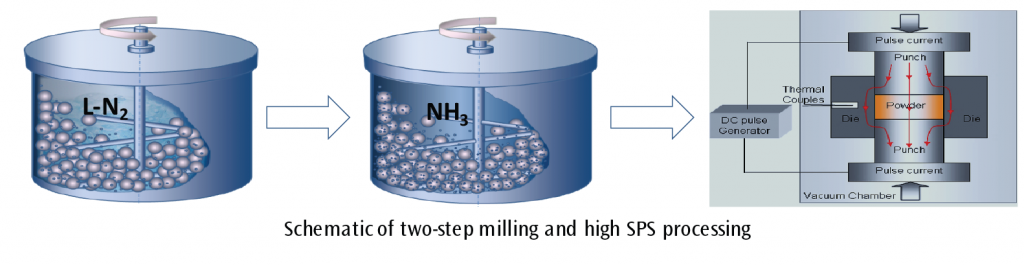
The first step in this novel synthesis process takes pure Fe powder (such as commercial gas atomized iron powder) and cryomills it using liquid nitrogen, producing a finer grained material with a significant amount of vacancies, grain boundaries and dislocations, allowing for faster diffusion of nitrogen gas into the iron crystalline lattice. Unlike typical Fe powder, cryomilled powder allows nitriding during the second ball-milling step with NH3 to occur at room temperature, eliminating the need for high temperatures. Finally, the raw iron nitride nanocrystalline powder is sintered to form dense iron nitrides (such as g’-Fe4N) using SPS, which prevents significant grain growth and decomposition of iron nitride phases due to its low sintering temperature (< 600 °C), fast heating rate, and brief consolidation cycle. The resulting product is produced in less time and with considerably less energy than conventional methods. Additionally, through the use of shaped dies, SPS can directly create parts using a process called net-shaping, which completely eliminates the need for follow-up machining. Parts, such as toroidal and E-core shaped transformer cores, can be fabricated directly from raw iron nitride powders.
- Cost effective
- Time-saving
- Environmentally-friendly
- Electronics/magnetics manufacturing
- Renewable energy
- Transportation
- Grid scale energy
- Distributed energy at the home/consumer level
- Defense
SD#13050
Published10/2/2015
Last Updated8/3/2018
