Quenching of Unincorporated Amplification Signal Reporters (QUASR)
Sandia’s QUASR modification to LAMP helps satisfy the requirements for a simple, robust, and inexpensive point-of-care deployed diagnostic device
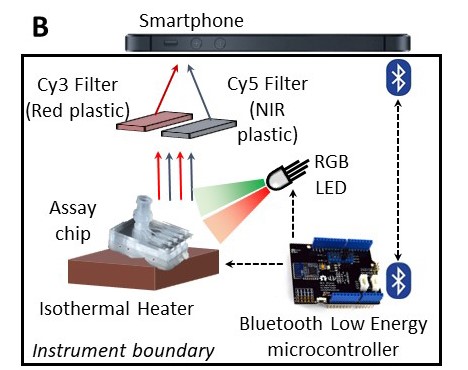
Rapid changes in how the world must react to pandemics has led to an unprecedented effort to develop rapid, easy to interpret point-of-care tests that meet the World Health Organization (WHO) standards. To address this critical need, Sandia National Laboratories developed QUASR (Quenching of Unincorporated Amplification Signal Reporters), a technique to significantly enhance the favorable characteristics of LAMP (Loop-mediated Isothermal Amplification). Combining these capabilities enables SmartLAMP, a smartphone-based deployable diagnostic device to create a simple, inexpensive, and robust system to quickly assess large and potentially dangerous public health situations.
The basics of Sandia’s diagnostic technology is founded on LAMP, a primer-based amplification of DNA/RNA targets. LAMP is one of several isothermal techniques with numerous desirable characteristics and benefits, such as fast response (5-30 min), robust and simple design, high sensitivity, low capital cost, and low power requirements. It can also work with minimal to no sample pretreatment, which is ideal for deployed diagnostics. As a PCR alternative, LAMP has existed for over a decade, yet never became mainstream due to the potential for false positives and lack of multiplexing. Sandia has overcome these issues by incorporating its unique and novel approach to a primer design called QUASR. QUASR significantly brightens the endpoint signal in a closedtube detection LAMP assay, allowing for a visual difference between positive and negative results that can be discerned and documented with simple equipment like a digital camera, smartphone, or even with the naked eye using a colored filter.
- Massively reduced cost compared to current testing equipment
- Faster and more accurate testing of human samples – results within 30 minutes
- User-friendly – no need for expensive equipment and technical training
- Eliminates need for lab preparation of biological samples and refrigeration
- Device can be adapted to detect other human or animal pathogens
- Distinguishes current and future SARS-CoV-2 variants and pandemics
- Limited-resource clinics
- Areas with minimally trained medical professionals and/or limited facilities
- Any area that needs to detect human or animal pathogens

SD#13252
Published8/9/2021
Last Updated8/16/2021